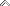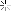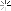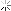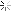Old Prius battery experiences
#11
Hi Andy,
Once I figured out that 04024 (SN: 03XBHB04024C) was not going to recover and seeing the wild voltage changes, I decided to use it for mechanical analysis and testing. That is why I cut off the top, to see the interior.
Later, I picked up some of the electrode fragments:

At the top you can see how the metal hydride electrode has a thin, mesh that covers it to keep it separated from the nickel hydroxide electrode. There is another example at the bottom where the metal hydride has slipped out of the separator. The patents describe this as a plastic material.
I suspect that as the water leaves the cell, the active area of the cell electrodes decreases. Meanwhile, the electrical loads do not. This leads to ohmic and some chemical heating in that smaller area with higher, spot temperatures. Eventually, the separator material gets so hot that it melts and no longer keeps the electrodes separated . . . a shorted cell. This can not be repaired.
My thinking is the best way to avoid this failure mode is to keep the cells well hydrated so the maximum area is working. This avoids a local hot-spot that fails the cell. It is also why I suspect that as long as a cell has not shorted, re-hydration not only restores them to service but significantly extends their life.
Bob Wilson
ps. If there were a fiber separator with very high temperature resistance, eventually all of the water would leave the cell and it would be inert. However, adding water would restore full capability.
Later, I picked up some of the electrode fragments:

At the top you can see how the metal hydride electrode has a thin, mesh that covers it to keep it separated from the nickel hydroxide electrode. There is another example at the bottom where the metal hydride has slipped out of the separator. The patents describe this as a plastic material.
I suspect that as the water leaves the cell, the active area of the cell electrodes decreases. Meanwhile, the electrical loads do not. This leads to ohmic and some chemical heating in that smaller area with higher, spot temperatures. Eventually, the separator material gets so hot that it melts and no longer keeps the electrodes separated . . . a shorted cell. This can not be repaired.
My thinking is the best way to avoid this failure mode is to keep the cells well hydrated so the maximum area is working. This avoids a local hot-spot that fails the cell. It is also why I suspect that as long as a cell has not shorted, re-hydration not only restores them to service but significantly extends their life.
Bob Wilson
ps. If there were a fiber separator with very high temperature resistance, eventually all of the water would leave the cell and it would be inert. However, adding water would restore full capability.
Last edited by bwilson4web; 03-06-2008 at 01:49 AM.
#12
Thanks again Bob,
I would agree with your explanation.
So the failed cells had no electrolye solution left whatsoever? Or enough of the separator was exposed that it was not cooled. The poor efficiency of the used cells 30-60% shows that there is a non-conductive/high resistance film present on the electrodes which will heat the electrolyte solution leading to water blow off from the pressure release valves. Keeping the electrolyte topped up should prevent a "hard" failure. I think you would still need periodic reconditioning/restore by the method you showed to extend the life of the battery.
Great work
Thanks
Andrew
I would agree with your explanation.
So the failed cells had no electrolye solution left whatsoever? Or enough of the separator was exposed that it was not cooled. The poor efficiency of the used cells 30-60% shows that there is a non-conductive/high resistance film present on the electrodes which will heat the electrolyte solution leading to water blow off from the pressure release valves. Keeping the electrolyte topped up should prevent a "hard" failure. I think you would still need periodic reconditioning/restore by the method you showed to extend the life of the battery.
Great work
Thanks
Andrew
#13
. . . present on the electrodes which will heat the electrolyte solution leading to water blow off from the pressure release valves. Keeping the electrolyte topped up should prevent a "hard" failure. I think you would still need periodic reconditioning/restore by the method you showed to extend the life of the battery. . . .
I don't have enough information about electrode activation but I suspect there is a layer of gas, probably oxygen, on the surface that has to be 'purged' so the KOH can reach the metal hydride. This layer would prevent the hydrogen molecules being absorbed into the hydride matrix. I suspect electrode activation applies enough charge to displace and/or generate enough hydrogen to convert the surface oxygen into hydride ions.
I am not convenced that battery reconditioning, the use of a .1C or lower charge, is as effective as claimed. The reason is I have some AA NiMH batteries that had excellent capacity, 2,000 mAhr, that after using in my digital camera now show barely 800 mAhr. I suspect excessive currents have led to premature, out-gassing and loss of capacity. Over in Prius Technical Stuff, I'm planning to do the test as well as putting in some probes to measure individual cell characteristics.
Bob Wilson
Last edited by bwilson4web; 03-08-2008 at 05:30 AM.
Thread
Topic Starter
Forum
Replies
Last Post